Continental Clinical Research
Cutting-edge Solutions for Medical Research.
Delivering quality data, efficient execution, and ethical excellence.
Years of Clinical Research experience
%
Historical Retention Rate
Clinical trials completed in last 3 years
%
GCP/ICH Trained Staff
Built for Modern Clinical Trials
Empowering Healthcare Innovation Trough Precise Clinical Research
Continental Clinical Research is a United States based, high performance clinical research site built for modern clinical trials. We combined experienced, board certified physicians, specialized teams, and an integrated research infrastructured.
We partner with sponsor, CROs, and biotech companies to deliver reliable data, rapid patient enrollment, and efficient trial execution under the highest ethical and regulatory standards.
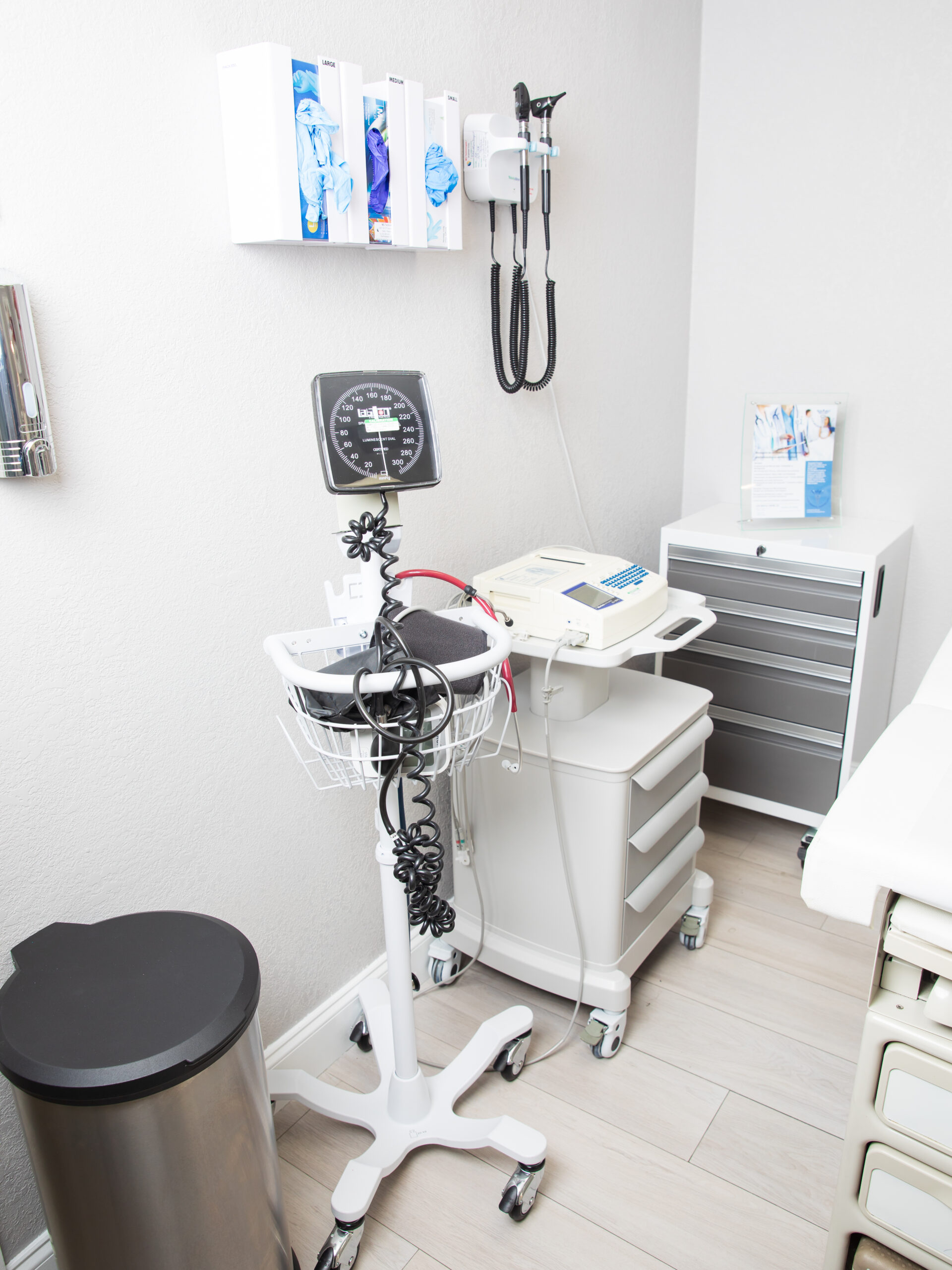
Our State of the Art and Modern facility
- Dedicated exam rooms
- IP storage ( temp logs , restricted acces)
- ECG, vitals, spirometry , etc.
- Refrigerated & ambient sample storage
- Calibration & maintenance SOPs
- Emergency equipment
- Multiple EDC platform familiarity
- Remote monitoring capabilities
- CCTV 24-7
- Cloud based and physical back up of all data
- Building with central electric generator for emergencies
High Recruitment Capabilities
- Active databases
- Community outreach programs
- Local physicians partnerships
- Digital recruitment strategy
- Optimal time from feasibility to first patient in (FPI)
Professional Staff
Experienced clinical trial team composed of dedicated coordinators, research nurses, data managers and regulatory specialists. Skilled in participant screening, informed consent, protocol compliance, and accurate data collection. Proficient in managing logistics, regulatory documentation, and participant follow up ensuring efficient trial execution, high recruitment and retention, and adherence to ethical and quality standards.
Our Core Metrics
With a robust regulatory framework aligned with internacional standards we at CCR operates under globally accepted guidelines ( e.g., Internacional Council for Harmonisation of Technical Requirements for Pharmaceuticals for Humans Use-ICH, and its ICH E6 (R3) Good Clinical Practice), ensuring ethical oversight, participant safety, data integrity, and regulatory compliance for trials across regions.

Average Randomization Time Less Than 5 DAYS.
CCR is always optimizing this metric to demostrate rapid screening to assignment cycle time, streamlined workflows, and strong operational readiness enabling timely trial enrollment and efficient study progress.

High Enrollment Rate
High enrollment rates at a clinical research site reflect strong participant outreach, efficient screening and coordination, and effective site staff engagement all of which support robust data collection and timely trial completion. At CCR we are committed to keep that metric in optimal range.
Historical Retention Rate 99%
We have exceptional participant engagement and follow through, reflecting strong site participant relationship, robust retention strategies, and reliable data completeness all of which bolster study integrity and credibility.
Principal Investigator

Juan Carlos Gutierrez MD
Internal Medicine
A seasoned clinical research physician with over 14 years of experience conducting Phase II–IV clinical trials across multiple therapeutic areas. Earned a medical degree from Universidad de Ciencias Médicas de La Habana, Cuba, and completed a Rotatory Internship at Hospital Buen Samaritano in Aguadilla, Puerto Rico.
Board-certified in Family Medicine, he combines strong clinical expertise with proven research leadership. He has successfully led high-performing teams, demonstrated excellence in patient recruitment and trial execution, and consistently delivered high-quality data while maintaining strict regulatory and ethical standards.
Certifications
• ACLS (2021)
• PALS (2021)
• BLS (2021)
• GCP (Updated)
Languages
Spanish, English, Portuguese
Clinical Trial Experience
Infectious Diseases
• COVID-19
• Clostridioides difficile infection
• RSV (Respiratory Syncytial Virus)
• UTI Prevention Vaccine
Respiratory & Pulmonary
• COPD
Endocrinology & Metabolic
• Thyroid Eye Disease
Major Sponsors & CROs:
• Gilead Sciences
• Janssen
• Shionogi
• Viridian Therapeutics
• Chiesi Farmaceutici
IVRS/IWRS
- Perceptive
- Clinphone
- Rapids
- Bracket
- Impala
- Sharp
- 1360
EDC
- Oracle
- Inform
- Medrio
- Datalabs
- Medidata/Rave
- WebCRF
IRB
- Advarra
- WCG
Central Laboratories
- PPD
- Covance
- Lab Cop
- Quintiles
- Quest Diagnostics
Local Laboratories
- CM Labs
- First Quality Laboratories
- Finlay Labs
- Florida Reference Labs
Additional Training/Certifications
- Annual SOP review performed
- Annual GCP/ICH training
- Annual HIPPA and OSHA training
- Bi-annual CPR and AED/ACLS training
- IATA Certified
Why Choose Continental Clinical Research
High performance clinical research site with over 10 years of experience, delivering reliable data, efficient execution, and strong participant retention across multiple therapeutics areas.
What Makes Our Research Site Different
We combine experienced clinical professional, strong recruitment capabilities, and streamlined processes to ensure rapid enrollment, high retention, and reliable study outcomes.
